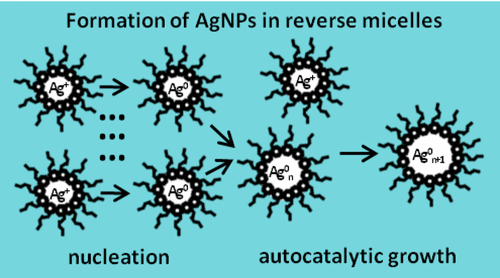
My master’s thesis and my PhD thesis were dedicated to studying the formation of gold and silver nanoparticles in reverse micellar solutions. The kinetics of this process were investigated at a quantitative level using UV-vis spectroscopy.
Kinetic mechanism includes the steps of slow, continuous nucleation and fast, autocatalytic surface growth. Both steps are under kinetic control of the precursor reduction. The rate of nucleation is limited by reaction in the droplets of the aqueous phase forming the cores of reverse micelles, and growth rate is limited by the reaction on the surface of particles growing inside the micelles. A detailed chemical mechanism for the reduction of the metal precursor was suggested for each studied system.
Peer-reviewed papers
Sergievskaya A.P., Tatarchuk V.V., Makotchenko E.V., Mironov I.V. Formation of gold nanoparticles during
the reduction of HAuBr4 in reverse micelles of oxyethylated surfactant: Influence of gold precursor
on the growth kinetics and properties of the particles // J.M.R. 2015. V. 30(12). P. 1925-1933. doi.org/10.1557/jmr.2015.121
Tatarchuk V.V., Sergievskaya A.P., Korda T.M., Druzhinina I.D., Zaikovsky V.I. Kinetic factors in the synthesis of silver nanoparticles by reduction of Ag+ with hydrazine in reverse micelles of Triton N-42 // Chem. Mater. 2013. V. 25(18). P. 3570-3579. doi.org/10.1021/cm304115j
Tatarchuk V.V., Sergievskaya A.P., Druzhinina I.A., Zaikovsky V.I. Kinetics and mechanism of the growth of gold nanoparticles by reduction of tetrachloroauric acid by hydrazine in Triton N-42 reverse micelles // J. Nanopart. Res. 2011. V. 13(10). P. 4997-5007. doi.org/10.1007/s11051-011-0481-1
Tatarchuk V.V., Sergievskaya A.P., Bulavchenko A.I., Zaikovsky V.I., Druzhinina I.A., Korda T.M., Gevko P.N., Alexeyev A.V. Di-(2-ethylhexyl) dithiophosphoric acid surface protected gold nanoparticles: micellar synthesis, stabilization, isolation and properties // Gold Bulletin. 2011. V. 44(4). P. 207-215. doi.org/10.1007/s13404-011-0031-8
Tatarchuk V.V., Bulavchenko A.I., Druzhinina I.A., Sergievskaya A.P. Effect of dioctyl sulfide on the kinetic of oxidative dissolution of gold nanoparticles in Triton N-42 reverse micelles // Russ. J. Inorg. Chem. 2011. V. 56(5). P. 808-815. doi.org/10.1134/S0036023611050251